Peer Reviewed Articles on Why Exercise Is Important
Introduction
Many evidences demonstrated that physical do (PE) affects brain plasticity, influencing cognition and wellbeing (Weinberg and Gould, 2015; for review run across Fernandes et al., 2017). In fact, experimental and clinical studies have reported that PE induces structural and functional changes in the brain, determining enormous biological, and psychological benefits.
In general, when reported PE effects, it is customary to dissever the biological aspects from the psychological ones. In fact, almost of the studies documented either the effects of PE on the brain (then on the cognitive functioning) or on the wellbeing (in terms of physical and mental health). In this review, we merge both these aspects every bit they influence each other. In fact, behaviorally advisable choices depend upon efficient cognitive functioning. Furthermore, emotional states influence cognitive functions through specific cerebral circuitry involving prefrontal areas and limbic structures (Barbas, 2000).
Before analyzing the benefits of PE, it is necessary to ascertain PE precisely. Indeed, PE is a term often incorrectly used interchangeably with concrete activeness (PA) that is "any bodily move produced by skeletal muscles that requires energy expenditure" (World Wellness Organization, 2010). And so, PA includes any motor behavior such as daily and leisure activities and information technology is considered a determinant lifestyle for general health status (Burkhalter and Hillman, 2011). Instead, PE is "a sub nomenclature of PA that is planned, structured, repetitive, and has as a last or an intermediate objective the improvement or maintenance of one or more than components of concrete fettle" (Globe Health Organisation, 2010). Examples of PE are aerobic and anaerobic action, characterized by a precise frequency, duration and intensity.
In this review, we illustrate the biological and psychological benefits of PE on cognition and wellbeing both in health and diseases, reporting data from both beast and human being studies. The biological footing at both molecular and supramolecular level have been largely studied. The other aim of present piece of work is to written report the actual evidence on the epigenetic mechanisms that decide or modulate the biological furnishings of PE on the brain. In fact, while the biologic mechanisms are sufficiently studied both at the molecular and supramolecular levels (see Lista and Sorrentino, 2010), little is known about the epigenetic ones. Finally, the modality with which PE should be practiced to gain such advantages while avoiding negative consequences volition be discussed. In Table one are reported the inclusion and exclusion criteria for studies discussed in this review.

Table 1. Inclusion and exclusion criteria for studies included in this review.
Physical Practice, Brain, and Cognition
Amongst the biological furnishings of PE, those linked to "neuroplasticity" are quite important.
Neuroplasticity is an important feature of the nervous system, which can alter itself in response to experience (Bavelier and Neville, 2002). For this reason, PE may be considered as an enhancer environmental factor promoting neuroplasticity.
In animal studies, the structural changes analyzed concern the cellular (neurogenesis, gliogenesis, synaptogenesis, angiogenesis) and molecular (amending in neurotransmission systems and increasing in some neurotrophic factors) level (Gelfo et al., 2018), while the functional activity has been measured using the levels of performance in behavioral tasks, such every bit spatial tasks that allow to clarify the different facets of spatial cognitive functions (Mandolesi et al., 2017). In humans, indicators of structural changes correspond for instance to brain volumes, measures of white affair integrity or modulation in neurotrophins levels (by correlation with trophic factors plasma levels). Such metrics can be correlated to cognitive performances, defining the functional neural efficiency (Serra et al., 2011). To this regard, information technology should be emphasized that any morphological modify results in a modification of the functional properties of a neural circuit and vice versa any change in neuronal efficiency and functionality is based on morphological modifications (Mandolesi et al., 2017).
Experimental and clinical studies take shown that PE induces important structural and functional changes in brain functioning. In Tabular array two are reported the more evident effects induced by PE.

Table 2. Structural and functional effects of PE.
Animal Studies
In animals, motor activity or motor exercise are terms often used instead of PE. The effects of motor practice are mainly studied in rodents past means of specific training on wheels or by locomotor activeness analyses.
Studies on healthy animals take demonstrated that intense motor action increases neurons and glia cells proliferation rates in the hippocampus and the neocortex (van Praag et al., 1999a,b; Dark-brown et al., 2003; Ehninger and Kempermann, 2003; Steiner et al., 2004; Hirase and Shinohara, 2014) and induces angiogenesis in the neocortex, hippocampus, and cerebellum (Black et al., 1990; Isaacs et al., 1992; Kleim et al., 2002; Swain et al., 2003; Ekstrand et al., 2008; Gelfo et al., 2018). At the molecular level, motor activity causes changes in neurotrasmitters such as serotonin, noradrenalin, and acetylcholine (Lista and Sorrentino, 2010; for a review, see Lin and Kuo, 2013) and induces the release of the brain-derived neurotrophic gene (BDNF Vaynman et al., 2004; Lafenetre et al., 2011) and the insulin-like growth factor-1 (IGF-1; for a review, van Praag, 2009).
Animals performing motor exercise showed improvements in spatial abilities (van Praag et al., 2005; Snigdha et al., 2014) and in other cognitive domains such as executive functions (Langdon and Corbett, 2012), evidencing thus that motor exercise amend cognitive functions.
Similar structural and functional changes were evident even in older animals (Kronenberg et al., 2006) and in fauna models of neurodegenerative diseases (Nithianantharajah and Hannan, 2006), suggesting that motor practice is a potent neuroprotective factor against physiological and pathological aging (Gelfo et al., 2018). In this context, i can use transgenic models to determine exactly when a structural alteration occurs, and and then to study when the animals should undergo motor preparation in order to maximize its furnishings. To this regard, converging evidence is showing that motor activeness should be performed before the development of neurodegeneration in gild to exert its protective role (Richter et al., 2008; Lin et al., 2015) such as before the formation of beta amyloid plaques in Alzheimer'south illness (Adlard et al., 2005). However, there are some experimental evidences showing that motor exercise performed after neurodegenerative lesions permits to better spatial abilities, hence being also a strong therapeutic agent (Sim, 2014; Ji et al., 2015).
Interestingly, PE induces modifications that can be passed on to the offspring. In fact, positive maternal experiences can influence the offspring at both behavioral and biochemical levels (see Cutuli et al., 2017, 2018). Preclinical studies likewise indicated that the effects of maternal exercise during pregnancy can be passed on to offspring (Robinson et al., 2012). However, it is not clear if the possibilities of inheritance are limited to motor exercise alone. To this regard, information technology has been seen that meaning rats exposed to motor exercise on bicycle-running and treadmill running have offspring with improved spatial memory, and increased hippocampal BDNF level (Akhavan et al., 2008; Aksu et al., 2012). Withal, further studies are necessary since it remains unclear whether these beneficial effects upshot from physiological changes to the in utero environment and/or from epigenetic modifications to the developing embryo (Brusk et al., 2017). On the other hand, few studies, conflicting and hard to replicate, do non yet allow to explore the transgenerational effects of paternal motor exercise (Curt et al., 2017).
Human Studies
Neuroplasticity phenomena post-obit PE have been evidenced fifty-fifty in humans. A bully number of studies demonstrated that in adults, PE determines structural changes such as increased gray affair volume in frontal and hippocampal regions (Colcombe et al., 2006; Erickson et al., 2011) and reduced damage in the gray matter (Chaddock-Heyman et al., 2014).
Moreover, PE facilitates the release of neurotrophic factors such as peripheral BDNF (Hötting et al., 2016), increases blood menstruum, improves cerebrovascular wellness and determines benefits on glucose and lipid metabolism conveying "food" to the brain (Mandolesi et al., 2017).
These furnishings are reflected on cognitive operation (for a review see Hötting and Röder, 2013). In fact, the results of cross-sectional and epidemiological studies showed that PE enhances cerebral functions in young and older adults (Lista and Sorrentino, 2010; Fernandes et al., 2017), improving memory abilities, efficiency of attentional processes and executive-control processes (Kramer et al., 1999; Colcombe and Kramer, 2003; Grego et al., 2005; Pereira et al., 2007; Winter et al., 2007; Chieffi et al., 2017). Furthermore, structural changes following PE have been related to academic achievement in comparison to sedentary individuals (Lees and Hopkins, 2013; Donnelly et al., 2016). In this line, it has been also showed that children who practice regular aerobic activeness performed better on verbal, perceptual and arithmetic exam in comparison to sedentary ones of aforementioned age (Sibley and Etnier, 2003; Voss et al., 2011).
Numerous studies take demonstrated that PE prevents cognitive decline linked to aging (Yaffe et al., 2009; Hötting and Röder, 2013; Niemann et al., 2014), reduces the adventure of developing dementia (Colberg et al., 2008; Mandolesi et al., 2017), the level of deterioration in executive functions (Hollamby et al., 2017) and improves the quality of life (Pedrinolla et al., 2017). Furthermore, positron emission tomography based studies evidenced that PE determines changes in metabolic networks that are related to knowledge (Huang et al., 2016).
Recently, studies on magnetoencephalography based (MEG) functional connectivity evidenced that PE influences network topology (Foster, 2015). Information technology is important to underlie that MEG is a much more direct measure of neural activity in comparison to fRMI, with the advantage of combining good spatial and high temporal resolution. In healthy individuals, PE was related to ameliorate intermodular integration (Douw et al., 2014) and to improvements in cognitive functions (Huang et al., 2016). Benefits of PE are evidenced even in individuals at risk for AD (Deeny et al., 2008), thus once over again suggesting a protective role of PE.
A possible explanation for these ameliorative structural and functional effects could be that PE stimulates claret circulation in the neural circuits involved in cerebral operation (Erickson et al., 2012). Another estimation could be found in the concept of "cerebral reserves" (Stern, 2002, 2012) a mechanisms that might explain why, in the face of neurodegenerative changes that are similar in nature and extent, individuals vary considerably in the severity of cognitive aging and clinical dementia (Petrosini et al., 2009). Two types of reserves are recognized: brain reserve and cognitive reserve. The erstwhile is based on the protective potential of anatomical features such equally brain size, neuronal density and synaptic connectivity, the latter is based on the efficient connectivity among neural circuits (Stern, 2002; Mandolesi et al., 2017).
According to the reserves hypothesis and taking into business relationship the numerous evidences described above, we could advance that PE is an environmental factor that permits to proceeds reserves.
However, i must underline that if on the i paw PE improves the cognitive performance, providing reserves to be spent in the case of a brain lesion, on the other hand the modifications of the clinical expression of neurodegeneration delays the diagnosis. It has been seen that patients with higher cognitive reserve take longer to manifest the symptoms of memory loss (Zanetti et al., 2017). It has been hypothesized a neural compensation mechanism that permits to perform complex activities (Stern, 2009). Manifestly, these conclusions open important reflections more for the diagnosis of neurodegenerative disease than for the practice of PE.
The effects of PE on cognitive performance accept been shown beyond the lifespan from childhood to the one-time age (Hötting and Röder, 2013). In item, information technology has been evidenced that cerebral functions that are influenced the most by brain maturation, such as attending or cerebral flexibility, and the cognitive functions that depend the most upon experiences, such as retention, are the most sensitive ones to PE (Hötting and Röder, 2013). Overall, these studies, together with those analyzing the effects of combined environmental factors, suggest that for a positive outcome on cerebral function, it is necessary to maintain an "enriched lifestyle" upwards to middle life. In fact, the exposure to PE together to other many experiences provides a "reserve"-like reward which supports an enduring preservation of cognitive function in erstwhile historic period (Chang et al., 2010; Loprinzi et al., 2018).
Concrete Practise and Wellbeing
There are consistent evidences that PE has many benefits for people of whatever age, improving psychological wellbeing (Zubala et al., 2017) and quality of life (Penedo and Dahn, 2005; Windle et al., 2010; Table 3).

Table 3. Biological and psychological effects of PE (Adapted from Weinberg and Gould, 2015).
In children, PE is correlated with high levels of self-efficacy, tasks goal orientation, and perceived competence (Biddle et al., 2011). In youth and adulthood, most studies evidenced that PE is associated with better health outcomes, such as better mood and self-concept (Berger and Motl, 2001; Landers and Arent, 2001; Penedo and Dahn, 2005). In the aging population, PE helps maintaining independence (Stessman et al., 2009), favoring social relations and mental health.
It was now well-accepted that is the interaction between biological and psychological mechanisms linked to PE enhances the wellbeing (Penedo and Dahn, 2005). Biological mechanisms of beneficial furnishings of PE are mainly related to increasing in cerebral blood flow and in maximal oxygen consumption, to delivery of oxygen to cognitive tissue, to reduction in musculus tension and to increased serum concentrations of endocannabinoid receptors (Thomas et al., 1989; Dietrich and McDaniel, 2004; Querido and Sheel, 2007; Gomes da Silva et al., 2010; Ferreira-Vieira et al., 2014). Moreover, neuroplasticity phenomena such every bit changes in neurotransmitters are recognized to bear on wellbeing. For instance, PE increases the levels of serotonin (Immature, 2007; Korb et al., 2010) and the levels of beta-endorphins, such as anandamide (Fuss et al., 2015).
Amongst the psychological hypothesis proposed to explain how PE enhances wellbeing, information technology has been underlined feeling of control (Weinberg and Gould, 2015), competency and self-efficacy (Craft, 2005; Rodgers et al., 2014), improved self-concept and self-esteem (Marsh and Sonstroem, 1995; Flim-flam, 2000; Zamani Sani et al., 2016), positive social interactions and opportunities for fun and enjoyment (Raedeke, 2007; Bartlett et al., 2011).
Psychological research evidenced that PE can even modulate the personality and the development of Cocky (Weinberg and Gould, 2015). Moreover, PE has been correlated with hardiness, a personality way that enables a person to withstand or cope with stressful situations (Weinberg and Gould, 2015).
In the following sections, we volition focus on correlations amidst PE and the most mutual mental illnesses.
Depression and Anxiety
Depression is the most common type of mental illness and will be the second leading cause of disease by 2020 (Farioli-Vecchioli et al., 2018). Similar entity concerns anxiety disorders that are amongst the most prevalent mental disorders in the earth population (Weinberg and Gould, 2015).
Epidemiological studies accept consistently reported benefits of PE on reductions in low (Mammen and Faulkner, 2013) and anxiety (DeBoer et al., 2012). For example, it has been seen that individuals that do PE regularly are less depressed or anxious than those who do not (De Moor et al., 2006), suggesting the employ of exercise equally a treatment for these illnesses (Carek et al., 2011).
Nearly of the enquiry on the relationship between PE and positive changes in mood state has evidenced positive effects, especially as a consequence of aerobic exercise, regardless of the specific blazon of activeness (Knapen et al., 2009), fifty-fifty if the correct intensity of aerobic PE to control and reduce symptoms is debated (de Souza Moura et al., 2015). For example, information technology has been revealed that subsequently about 16 weeks of an aerobic exercise program, individuals with major depressive disorder (MDD), significantly reduced their depressive symptoms (Arts and crafts and Perna, 2004). However, there are evidenced that documented that fifty-fifty anaerobic activeness has positive effects on treatment of clinical low (Martinsen, 1990). For feet disorders, it has been evidenced that the positive effects of PE are visible fifty-fifty with short bursts of exercise, independently from the nature of the exercise (Scully et al., 1998).
A physiologic machinery correlated to the improvement in depressed mood mail-exercise PE was identified in modulation of peripheral levels of BDNF (Coelho et al., 2013). In this line, it was suggested recently that the intensity of practice to improve mood should be prescribed on individual basis and not on the patient'due south preferred intensity (Meyer et al., 2016a,b). Conversely, physical inactivity correlated to worse depressive symptoms and, then, to lower peripheral levels of BDNF (Brunoni et al., 2008). Post-PE mood comeback might likewise be due to lower oxidative stress (Thomson et al., 2015). In this contest, it was evidenced that at that place is an aberrant oxidative stress in individuals with MDD or bipolar disorder (Cataldo et al., 2010; Andreazza et al., 2013) and that PE, especially in higher intensity, decreases oxidative stress with consequent mood improvement (Urso and Clarkson, 2003).
Addictive and Unhealthy Behaviors
PE has been widely evidenced to exist an constructive tool for treating several addictive and unhealthy behaviors. PE tends to reduce and prevent behaviors such equally smoking, alcohol, and gambling, and to regulate the impulse for hunger and satiety (Vatansever-Ozen et al., 2011; Tiryaki-Sonmez et al., 2015). In this context, several studies evidenced substance abusers benefit from regular PE, that also helps increasing good for you behaviors (Giesen et al., 2015). Information technology has been evidenced that regular PE reduces tobacco cravings and cigarette employ (Haasova et al., 2013). Although PE has positive effects on psychological wellbeing, in this context it is right underline that in some cases PE could reveal unhealthy behaviors with negative consequence on health (Schwellnus et al., 2016). It is the case of practice addiction, a dependence on a regular regimen of practice that is characterized by withdrawal symptoms, after 24–36 h without practice (Sachs, 1981), such every bit feet, irritability, guilt, muscle twitching, a bloated feeling, and nervousness (Weinberg and Gould, 2015). At that place is a strong correlation between practise addiction and eating disorders (Scully et al., 1998) suggesting thus a comorbidity of these disorders and a mutual biological substrate. In particular, recent studies have shown that these unhealthy behaviors are associated to lower prefrontal cortex volume, activity and oxygenation, with consequent impairment in cognitive functions, such as the inhibitory command with the consistent compulsive behaviors (Asensio et al., 2016; Wang et al., 2016; Pahng et al., 2017). Also, it has been seen that a few days of PE increase oxygenation of prefrontal cortex, improving mental wellness (Cabral et al., 2017).
Epigenetic Mechanisms
Biological and psychological furnishings of PE could be partly explained through epigenetic mechanisms. The term "epigenetics," coined past Waddington (1939), is based on a conceptual model designed to account for how genes might interact with their environment to produce the phenotype (Waddington, 1939; Fernandes et al., 2017).
In particular, epigenetics is referred to all those mechanisms, including functional modifications of the genome such equally DNA methylation, post-translational histone modifications (i.east., acetylation and methylation) and microRNA expression (Deibel et al., 2015; Grazioli et al., 2017), which tend to regulate gene expression, modeling the chromatin structure but maintaining the nucleotide sequence of Dna unchanged.
The electric current literature conspicuously demonstrates that these mechanisms are strongly influenced by unlike biological and environmental factors, such equally PE (Grazioli et al., 2017), which determine the nature and the mode of epigenetic mechanisms activation.
Epigenetics plays an essential role in neural reorganization, including those that govern the brain plasticity (Deibel et al., 2015). For case, a growing body of evidence indicates that regulates neuroplasticity and memory processes (Ieraci et al., 2015).
Several animal studies reveal how motor activity is able to ameliorate cerebral performances acting on epigenetic mechanisms and influencing the expression of those genes involved in neuroplasticity (Fernandes et al., 2017). The master molecular processes that underlie the epigenetic mechanisms are the following: through DNA methylation, histone modifications and microRNA expression (Fernandes et al., 2017).
Deoxyribonucleic acid methylation is a chemical covalent modification on the cytosine of the double stranded DNA molecule. It has been recognized that Dna methylation plays a key function in long-term retention (Deibel et al., 2015; Kim and Kaang, 2017). In detail, mechanisms related to DNA methylation save the repressive effects of memory-suppressor genes to favor the expression of plasticity-promoting and memory consolidation genes. Several evidences showed that PE is able to coordinate the action of the genes involved in synaptic plasticity that regulate memory consolidation (Molteni et al., 2002; Ding et al., 2006).
Histone modifications are post-translational chemical changes in histone proteins. They include histone methylation/demethylation, acetylation/deacetylation, and phosphorylation, all due to the action of specific enzymes, which modify the chromatin construction, thereby regulating gene expression. It has been demonstrated that histone acetylation is a requisite for long-term memory (LTM) (Barrett and Wood, 2008; Fernandes et al., 2017). In animals, motor activity increases these genetic mechanisms in the hippocampus and the frontal cortex, improving memory performances in behavioral tasks (Cechinel et al., 2016). Recently, following iv weeks of motor exercise, it has been evidenced an increasing of the activity of enzymes involved in histone acetylation/deacetylation, the epigenetic mechanisms that make up one's mind an enhancing in the expression of BDNF (Maejima et al., 2018).
MicroRNAs (miRNAs) are minor, single stranded RNA molecules able to inhibit the expression of target genes. They are widely expressed in the brain, participating in epigenetic mechanisms and acting every bit regulators of numerous biological processes in the brain, ranging from jail cell proliferation, differentiation, apoptosis, synaptic plasticity, and retentivity consolidation (Saab and Mansuy, 2014). Recent evidences demonstrate that PE tin can mitigate the harmful furnishings of traumatic brain injury and crumbling on cerebral part by regulating the hippocampal expression of miR21 (Hu et al., 2015) and miR-34a (Kou et al., 2017). Furthermore, PE contributes to attenuate the effects of stress-related increase in miR-124, involved in neurogenesis and memory formation (Pan-Vazquez et al., 2015).
What Kind of Physical Exercise?
Sport psychology has suggested that the success or failure of PE programs depends on several factors such equally the intensity, frequency, elapsing of the exercise, and whether the PE is washed in group or alone (Weinberg and Gould, 2015). These aspects are important in terms of maintenance of PE practice and in order to proceeds benefits for brain and behavior, and they are affected by private characteristics. Although such aspects accept to be taken into account when preparation is proposed, scientific reports have evidenced different effects on cognitive performance and wellbeing if PE is performed in aerobic or anaerobic modality.
Aerobic practice allows the resynthesis of adenosine—triphosphate (ATP) by aerobic mechanisms, adjusting intensity (from low to high intensity), duration (normally long), and oxygen availability. The intensity depends on the cardiorespiratory effort with respect to the maximum heart rate (HRmax) or the maximum oxygen consumption (Vo2max), which determines an increment in oxygen consumption with respect to the residue condition. Examples of aerobic PE are jogging, running, cycling, and swimming.
On the contrary, anaerobic do has high intensity, short duration and unavailability of oxygen, determining the depletion of the muscles' ATP and/or phosphocreatine (PCr) reserves, shifting the production of ATP, to anaerobic energy mechanisms, lactacid or alactacid. Examples of anaerobic exercises are weight lifting or sprint in 100 one thousand.
Robust literature demonstrated that chronic aerobic exercise is associated with potent structural and functional neuroplastic changes, with an comeback in cognitive functions (Colcombe et al., 2006; Hillman et al., 2008; Erickson et al., 2009; Mandolesi et al., 2017) and increased feeling of general wellbeing (Berger and Tobar, 2011; Biddle et al., 2011) (Table 4).
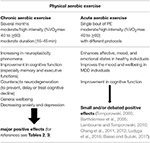
Tabular array iv. Effects of physical aerobic do on cognitive functioning and wellbeing.
Recently, growing show showed that astute aerobic practice, divers as a unmarried tour of exercise, relates to improved cognitive functions, especially prefrontal cortex-dependent cognition (Tomporowski, 2003; Lambourne and Tomporowski, 2010; Chang et al., 2011; Ludyga et al., 2016; Basso and Suzuki, 2017). All the same, the effects of a single session of exercise on cerebral functioning are generally small (Chang et al., 2012). In this line, it was evidenced that even a unmarried bout of moderate-intensity aerobic exercise enhances, mood and emotional states and improves the wellbeing in MDD individuals (Bartholomew et al., 2005; Basso and Suzuki, 2017) (Table 4).
Beside frequency and duration over fourth dimension, even the intensity is a parameter to be considered when evaluating the PE effects. It has been showed that moderate intensity exercise is related to increased performance in working memory and cognitive flexibility, whereas high-intensity exercise improves the speed of information processing (Chang and Etnier, 2009). In this context, it has been reported that peripheral BDNF was significantly increased after high intensity exercise, but not subsequently depression-intensity exercise (Hötting et al., 2016). In fact, information technology is evidenced that high-intensity exercise provides greater benefit to cerebral functions than low-intensity exercise in the elderly (Dark-brown et al., 2012).
With regard to the psychological beneficial effects related to PE, inquiry has evidenced that major benefits in reduction of anxiety and depression are adamant by longer grooming programme (several months), every bit compared to shorter ones (some days) for training session lasting over 30 min. Moreover, anxiety and depression reduction later aerobic do may be accomplished with exercise intensity between xxx and 70% of maximal heart charge per unit (Weinberg and Gould, 2015). To achieve positive mood changes, an of import role is played fifty-fifty by anaerobic activity, such as yoga, or in all PEs in which there is rhythmic abdominal breathing, enjoyment, rhythmic, and repetitive movements and relative absence of interpersonal competition (Berger and Motl, 2001).
Conclusion
PE determines positive biological and psychological effects that touch the brain and the cerebral operation and promote a condition of wellbeing. PE plays an important role in counteract normal and pathological aging. Recent evidences accept shown that PE triggers stiff neuroplastic phenomena, partly mediated past epigenetic mechanisms. In fact, PE crusade profound alterations in gene expression and its protein products in the form of epigenomic manifestations (Fernandes et al., 2017).
A growing torso of literature indicates that both chronic and aerobic PE can reach like benefits.
These results should atomic number 82 to reflect on beneficial furnishings of PE and to promote its employ as a modifiable cistron for prevention, to improve cognitive abilities and to enhance mood.
Despite all these positive effects, information technology must be underlined that PE should exist tailored to the individual. In fact, fifty-fifty PE, when excessive, can have a dark side, when PE becomes compulsive and facilitates addictive behaviors.
Writer Contributions
LM, AP, SM, FF, GF, PS, and GS: designed the review; LM and GS: wrote the newspaper. All authors read, revised, and approved the terminal manuscript.
Funding
The present paper was supported by University of Naples Parthenope Ricerca Competitiva 2017 (D.R. 289/2017) to LM and GF.
Conflict of Interest Statement
The authors declare that the enquiry was conducted in the absence of any commercial or financial relationships that could exist construed as a potential conflict of interest.
References
Adlard, P. A., Perreau, V. Yard., Popular, V., and Cotman, C. W. (2005). Voluntary exercise decreases amyloid load in a transgenic model of Alzheimer's disease. J. Neurosci. 25, 4217–4221. doi: x.1523/JNEUROSCI.0496-05.2005
PubMed Abstruse | CrossRef Total Text | Google Scholar
Akhavan, M. M., Emami-Abarghoie, Grand., Safari, M., Sadighi-Moghaddam, B., Vafaei, A. A., Bandegi, A. R., et al. (2008). Serotonergic and noradrenergic lesions suppress the enhancing effect of maternal exercise during pregnancy on learning and memory in rat pups. Neuroscience 151, 1173–1183. doi: ten.1016/j.neuroscience.2007.10.051
PubMed Abstruse | CrossRef Total Text | Google Scholar
Aksu, I., Baykara, B., Ozbal, Southward., Cetin, F., Sisman, A. R., Dayi, A., et al. (2012). Maternal treadmill practise during pregnancy decreases anxiety and increases prefrontal cortex VEGF and BDNF levels of rat pups in early on and late periods of life. Neurosci. Lett. 516, 221–225. doi: ten.1016/j.neulet.2012.03.091
PubMed Abstract | CrossRef Full Text | Google Scholar
Andreazza, A. C., Wang, J. F., Salmasi, F., Shao, L., and Immature, Fifty. T. (2013). Specific subcellular changes in oxidative stress in prefrontal cortex from patients with bipolar disorder. J. Neurochem. 127, 552–561. doi: 10.1111/jnc.12316
PubMed Abstruse | CrossRef Full Text | Google Scholar
Asensio, South., Morales, J. 50., Senabre, I., Romero, Thou. J., Beltran, M. A., Flores-Bellver, Grand., et al. (2016). Magnetic resonance imaging structural alterations in encephalon of alcohol abusers and its association with impulsivity. Aficionado. Biol. 21, 962–971. doi: ten.1111/adb.12257
PubMed Abstruse | CrossRef Total Text | Google Scholar
Barbas, H. (2000). Connections underlying the synthesis of knowledge, memory, and emotion in primate prefrontal cortices. Brain Res. Bull. 52, 319–330. doi: 10.1016/S0361-9230(99)00245-two
PubMed Abstract | CrossRef Total Text | Google Scholar
Barrett, R. K., and Wood, Grand. A. (2008). Across transcription factors: the part of chromatin modifying enzymes in regulating transcription required for memory. Learn. Mem. 15, 460–467. doi: x.1101/lm.917508
PubMed Abstract | CrossRef Full Text | Google Scholar
Bartholomew, J. B., Morrison, D., and Ciccolo, J. T. (2005). Effects of acute exercise on mood and well-being in patients with major depressive disorder. Med. Sci. Sport. Exerc. 37, 2032–2037. doi: 10.1249/01.mss.0000178101.78322.dd
PubMed Abstract | CrossRef Full Text | Google Scholar
Bartlett, J. D., Close, G. L., MacLaren, D. P. 1000., Gregson, Westward., Drust, B., and Morton, J. P. (2011). High-intensity interval running is perceived to be more enjoyable than moderate-intensity continuous exercise: implications for exercise adherence. J. Sports Sci. 29, 547–553. doi: 10.1080/02640414.2010.545427
PubMed Abstract | CrossRef Total Text | Google Scholar
Basso, J. C., and Suzuki, W. A. (2017). The effects of acute exercise on mood, noesis, neurophysiology and neurochemical pathways: a review. Encephalon Plast. 2, 127–152. doi: x.3233/BPL-160040
CrossRef Total Text | Google Scholar
Berger, B., and Motl, R. (2001). "Physical action and quality of life," in Handbook of Sport Psychology, eds R. N. Singer, H. A. Hausenblas, and C. Janelle (New York, NY: Wiley), 636–670.
Google Scholar
Berger, B., and Tobar, D. (2011). "Exercise and quality of life," in The NEW Sport and Practice Psychology Companion, eds T. Morris and P. Terry (Morgantown, WV: Fitness Information Technology), 483–505.
Biddle, S. J. H., Atkin, A. J., Cavill, N., and Foster, C. (2011). Correlates of physical activity in youth: a review of quantitative systematic reviews. Int. Rev. Sport Exerc. Psychol. four, 25–49. doi: 10.1080/1750984X.2010.548528
CrossRef Full Text | Google Scholar
Black, J. E., Isaacs, Thousand. R., Anderson, B. J., Alcantara, A. A., and Greenough, Due west. T. (1990). Learning causes synaptogenesis, whereas motor activeness causes angiogenesis, in cerebellar cortex of adult rats. Proc. Natl. Acad. Sci. U.s.a.A. 87, 5568–5572. doi: 10.1073/pnas.87.fourteen.5568
PubMed Abstruse | CrossRef Full Text | Google Scholar
Chocolate-brown, B. M., Peiffer, J. J., Sohrabi, H. R., Mondal, A., Gupta, V. B., Rainey-Smith, S. R., et al. (2012). Intense physical activity is associated with cognitive performance in the elderly. Transl. Psychiatry 2:e191. doi: 10.1038/tp.2012.118
PubMed Abstract | CrossRef Total Text | Google Scholar
Brown, J., Cooper-Kuhn, C. M., Kempermann, G., Van Praag, H., Winkler, J., Gage, F. H., et al. (2003). Enriched environment and physical action stimulate hippocampal but not olfactory bulb neurogenesis. Eur. J. Neurosci. 17, 2042–2046. doi: 10.1046/j.1460-9568.2003.02647.x
CrossRef Full Text | Google Scholar
Brunoni, A. R., Lopes, 1000., and Fregni, F. (2008). A systematic review and meta-analysis of clinical studies on major depression and BDNF levels: implications for the function of neuroplasticity in depression. Int. J. Neuropsychopharmacol. eleven, 1169–1180. doi: x.1017/S1461145708009309
PubMed Abstract | CrossRef Full Text | Google Scholar
Burkhalter, T. M., and Hillman, C. H. (2011). A narrative review of physical activity, nutrition, and obesity to cognition and scholastic performance across the human lifespan. Adv. Nutr. Int. Rev. J. 2, 201S−206S. doi: 10.3945/an.111.000331
PubMed Abstract | CrossRef Full Text | Google Scholar
Cabral, D. A., da Costa, K. Thousand., Okano, A. H., Elsangedy, H. 1000., Rachetti, V. P., and Fontes, E. B. (2017). Improving cerebral oxygenation, cognition and autonomic nervous organization command of a chronic alcohol abuser through a iii-calendar month running programme. Addict. Behav. Rep. 6, 83–89. doi: 10.1016/j.abrep.2017.08.004
PubMed Abstract | CrossRef Full Text | Google Scholar
Cataldo, A. M., McPhie, D. L., Lange, N. T., Punzell, S., Elmiligy, South., Ye, Due north. Z., et al. (2010). Abnormalities in mitochondrial structure in cells from patients with bipolar disorder. Am. J. Pathol. 177, 575–585. doi: 10.2353/ajpath.2010.081068
PubMed Abstract | CrossRef Total Text | Google Scholar
Cechinel, L. R., Basso, C. 1000., Bertoldi, K., Schallenberger, B., de Meireles, L. C. F., and Siqueira, I. R. (2016). Treadmill practise induces age and protocol-dependent epigenetic changes in prefrontal cortex of Wistar rats. Behav. Brain Res. 313, 82–87. doi: 10.1016/j.bbr.2016.07.016
PubMed Abstract | CrossRef Total Text | Google Scholar
Chaddock-Heyman, Fifty., Erickson, K. I., Holtrop, J. Fifty., Voss, 1000. Westward., Pontifex, Thou. B., Raine, Fifty. B., et al. (2014). Aerobic fitness is associated with greater white affair integrity in children. Front. Hum. Neurosci. eight:584. doi: 10.3389/fnhum.2014.00584
PubMed Abstract | CrossRef Full Text | Google Scholar
Chang, One thousand., Jonsson, P. V., Snaedal, J., Bjornsson, S., Saczynski, J. S., Aspelund, T., et al. (2010). The outcome of midlife concrete activeness on cerebral function among older adults: ages—Reykjavik study. J. Gerontol. Ser. A 65A, 1369–1374. doi: 10.1093/gerona/glq152
CrossRef Full Text | Google Scholar
Chang, Y. K., and Etnier, J. L. (2009). Exploring the dose-response human relationship between resistance exercise intensity and cognitive function. J. Sport Exerc. Psychol. 31, 640–656. doi: 10.1123/jsep.31.v.640
PubMed Abstract | CrossRef Full Text | Google Scholar
Chang, Y. K., Labban, J. D., Gapin, J. I., and Etnier, J. L. (2012). The furnishings of acute practice on cognitive performance: a meta-analysis. Brain Res. 1453, 87–101. doi: 10.1016/j.brainres.2012.02.068
PubMed Abstruse | CrossRef Full Text | Google Scholar
Chang, Y. G., Tsai, C. L., Hung, T. M., Then, East. C., Chen, F. T., and Etnier, J. L. (2011). Effects of acute exercise on executive function: a study with a tower of London job. J. Sport Exerc. Psychol. 33, 847–865. doi: ten.1123/jsep.33.6.847
PubMed Abstruse | CrossRef Full Text | Google Scholar
Chieffi, S., Messina, K., Villano, I., Messina, A., Valenzano, A., Moscatelli, F., et al. (2017). Neuroprotective effects of concrete activity: bear witness from human and animal studies. Front. Neurol. 8:188. doi: ten.3389/fneur.2017.00188
PubMed Abstract | CrossRef Total Text | Google Scholar
Coelho, F. M., de Gobbi, S., Andreatto, C. A. A., Corazza, D. I., Pedroso, R. 5., and Santos-Galduróz, R. F. (2013). Physical exercise modulates peripheral levels of brain-derived neurotrophic factor (BDNF): a systematic review of experimental studies in the elderly. Arch. Gerontol. Geriatr. 56, ten–15. doi: ten.1016/j.archger.2012.06.003
PubMed Abstract | CrossRef Full Text | Google Scholar
Colberg, S. R., Somma, C. T., and Sechrist, S. R. (2008). Physical activity participation may offset some of the negative touch on of diabetes on cerebral office. J. Am. Med. Dir. Assoc. nine, 434–438. doi: 10.1016/j.jamda.2008.03.014
PubMed Abstract | CrossRef Full Text | Google Scholar
Colcombe, S. J., Erickson, Thou. I., Scalf, P. Eastward., Kim, J. South., Prakash, R., McAuley, Due east., et al. (2006). Aerobic exercise training increases encephalon volume in aging humans. J. Gerontol. A Biol. Sci. Med. Sci. 61, 1166–1170. doi: x.1093/gerona/61.eleven.1166
PubMed Abstract | CrossRef Full Text | Google Scholar
Craft, Fifty. 50. (2005). Exercise and clinical low: examining two psychological mechanisms. Psychol. Sport Exerc. 6, 151–171. doi: 10.1016/j.psychsport.2003.11.003
CrossRef Full Text | Google Scholar
Cutuli, D., Berretta, Due east., Caporali, P., Sampedro-Piquero, P., De Bartolo, P., Laricchiuta, D., et al. (2018). Furnishings of pre-reproductive maternal enrichment on maternal care, offspring's play behavior and oxytocinergic neurons. Neuropharmacology. doi: ten.1016/j.neuropharm.2018.02.015. [Epub alee of print].
PubMed Abstract | CrossRef Full Text | Google Scholar
Cutuli, D., Berretta, E., Pasqualini, Yard., De Bartolo, P., Caporali, P., Laricchiuta, D., et al. (2017). Influence of pre-reproductive maternal enrichment on coping response to stress and expression of c-Fos and glucocorticoid receptors in adolescent offspring. Forepart. Behav. Neurosci. xi:73. doi: ten.3389/fnbeh.2017.00073
PubMed Abstract | CrossRef Total Text | Google Scholar
DeBoer, L. B., Powers, M. B., Utschig, A. C., Otto, Chiliad. Westward., and Smits, J. A. J. (2012). Exploring practise as an artery for the handling of anxiety disorders. Expert Rev. Neurother. 12, 1011–1022. doi: 10.1586/ern.12.73
PubMed Abstract | CrossRef Full Text | Google Scholar
Deeny, S. P., Poeppel, D., Zimmerman, J. B., Roth, South. Thou., Brandauer, J., Witkowski, S., et al. (2008). Exercise, APOE, and working memory: One thousand thousand and behavioral evidence for benefit of exercise in epsilon4 carriers. Biol. Psychol. 78, 179–187. doi: 10.1016/j.biopsycho.2008.02.007
PubMed Abstract | CrossRef Full Text | Google Scholar
Deibel, South. H., Zelinski, E. L., Keeley, R. J., Kovalchuk, O., and McDonald, R. J. (2015). Epigenetic alterations in the suprachiasmatic nucleus and hippocampus contribute to age-related cognitive decline. Oncotarget 6, 23181–23203. doi: 10.18632/oncotarget.4036
PubMed Abstract | CrossRef Full Text | Google Scholar
De Moor, Thou. H., Beem, A. L., Stubbe, J. H., Boomsma, D. I., and De Geus, E. J. C. (2006). Regular exercise, anxiety, depression and personality: a population-based study. Prev. Med. 42, 273–279. doi: x.1016/j.ypmed.2005.12.002
PubMed Abstract | CrossRef Full Text | Google Scholar
de Souza Moura, A. Chiliad., Lamego, Thousand. K., Paes, F., Rocha, N. B. F., Simoes-Silva, V., Rocha, South. A., et al. (2015). Comparison amidst aerobic practice and other types of interventions to treat depression: a systematic review. CNS Neurol. Disord. Drug Targets 14, 1171–1183. doi: 10.2174/1871527315666151111120714
PubMed Abstract | CrossRef Full Text | Google Scholar
Ding, Q., Vaynman, S., Akhavan, Chiliad., Ying, Z., and Gomez-Pinilla, F. (2006). Insulin-like growth gene I interfaces with brain-derived neurotrophic factor-mediated synaptic plasticity to modulate aspects of practise-induced cerebral function. Neuroscience 140, 823–833. doi: 10.1016/j.neuroscience.2006.02.084
PubMed Abstract | CrossRef Full Text | Google Scholar
Donnelly, J. E., Hillman, C. H., Castelli, D., Etnier, J. Fifty., Lee, South., Tomporowski, P., et al. (2016). Physical activity, fitness, cognitive function, and academic achievement in children. Med. Sci. Sport. Exerc. 48, 1197–1222. doi: ten.1249/MSS.0000000000000901
CrossRef Total Text | Google Scholar
Douw, L., Nieboer, D., van Dijk, B. Westward., Stam, C. J., and Twisk, J. W. (2014). A healthy brain in a salubrious body: brain network correlates of physical and mental fettle. PLoS ONE ix:e88202. doi: 10.1371/journal.pone.0088202
PubMed Abstract | CrossRef Total Text | Google Scholar
Ehninger, D., and Kempermann, M. (2003). Regional effects of wheel running and environmental enrichment on cell genesis and microglia proliferation in the adult murine neocortex. Cereb. Cortex 13, 845–851. doi: 10.1093/cercor/13.8.845
PubMed Abstract | CrossRef Full Text | Google Scholar
Ekstrand, J., Hellsten, J., and Tingström, A. (2008). Environmental enrichment, do and corticosterone affect endothelial jail cell proliferation in adult rat hippocampus and prefrontal cortex. Neurosci. Lett. 442, 203–207. doi: 10.1016/j.neulet.2008.06.085
PubMed Abstract | CrossRef Full Text | Google Scholar
Erickson, Chiliad. I., Miller, D. 50., Weinstein, A. M., Akl, S. Fifty., and Banducci, Due south. (2012). Physical activity and brain plasticity in late machismo: a conceptual and comprehensive review. Ageing Res. 3, 34–47. doi: ten.4081/ar.2012.e6
CrossRef Total Text | Google Scholar
Erickson, K. I., Prakash, R. S., Voss, M. W., Chaddock, L., Hu, Fifty., Morris, K. S., et al. (2009). Aerobic fettle is associated with hippocampal volume in elderly humans. Hippocampus 19, 1030–1039. doi: 10.1002/hipo.20547
PubMed Abstract | CrossRef Full Text | Google Scholar
Erickson, K. I., Voss, Thousand. Westward., Prakash, R. S., Basak, C., Szabo, A., Chaddock, Fifty., et al. (2011). Exercise training increases size of hippocampus and improves retentivity. Proc. Natl. Acad. Sci. U.S.A. 108, 3017–3022. doi: 10.1073/pnas.1015950108
PubMed Abstract | CrossRef Full Text | Google Scholar
Farioli-Vecchioli, S., Sacchetti, S., di Robilant, N. V., and Cutuli, D. (2018). The function of physical exercise and omega-3 fatty acids in depressive illness in the elderly. Curr. Neuropharmacol. 16, 308–326. doi: ten.2174/1570159X15666170912113852
PubMed Abstract | CrossRef Full Text | Google Scholar
Fernandes, J., Arida, R. M., and Gomez-Pinilla, F. (2017). Physical exercise as an epigenetic modulator of brain plasticity and cognition. Neurosci. Biobehav. Rev. 80, 443–456. doi: 10.1016/j.neubiorev.2017.06.012
PubMed Abstract | CrossRef Full Text | Google Scholar
Ferreira-Vieira, T. H., Bastos, C. P., Pereira, G. South., Moreira, F. A., and Massensini, A. R. (2014). A role for the endocannabinoid organisation in exercise-induced spatial memory enhancement in mice. Hippocampus 24, 79–88. doi: ten.1002/hipo.22206
PubMed Abstract | CrossRef Full Text | Google Scholar
Play tricks, K. R. (2000). Self-esteem, self-perceptions and exercise. Int. J. Sport Psychol. 31, 228–240.
Google Scholar
Fuss, J., Steinle, J., Bindila, L., Auer, M. K., Kirchherr, H., Lutz, B., et al. (2015). A runner's high depends on cannabinoid receptors in mice. Proc. Natl. Acad. Sci. U.S.A. 112, 13105–13108. doi: 10.1073/pnas.1514996112
PubMed Abstract | CrossRef Full Text | Google Scholar
Gelfo, F., Mandolesi, Fifty., Serra, L., Sorrentino, K., and Caltagirone, C. (2018). The neuroprotective effects of experience on cerebral functions: evidence from brute studies on the neurobiological bases of encephalon reserve. Neuroscience 370, 218–235. doi: 10.1016/j.neuroscience.2017.07.065
PubMed Abstract | CrossRef Full Text | Google Scholar
Giesen, Eastward. S., Deimel, H., and Bloch, W. (2015). Clinical practise interventions in alcohol utilise disorders: a systematic review. J. Subst. Corruption Treat. 52, 1–9. doi: 10.1016/j.jsat.2014.12.001
PubMed Abstract | CrossRef Full Text | Google Scholar
Gomes da Silva, S., Araujo, B. H. S., Cossa, A. C., Scorza, F. A., Cavalheiro, Due east. A., Naffah-Mazzacoratti Mda, M., et al. (2010). Concrete exercise in boyhood changes CB1 cannabinoid receptor expression in the rat brain. Neurochem. Int. 57, 492–496. doi: 10.1016/j.neuint.2010.07.001
PubMed Abstruse | CrossRef Full Text | Google Scholar
Grazioli, E., Dimauro, I., Mercatelli, Northward., Wang, G., Pitsiladis, Y., Di Luigi, L., et al. (2017). Concrete activity in the prevention of human diseases: role of epigenetic modifications. BMC Genomics 18:802. doi: 10.1186/s12864-017-4193-5
PubMed Abstruse | CrossRef Full Text | Google Scholar
Grego, F., Vallier, J. M., Collardeau, Thousand., Rousseu, C., Cremieux, J., and Brisswalter, J. (2005). Influence of do duration and hydration condition on cerebral function during prolonged cycling exercise. Int. J. Sports Med. 26, 27–33. doi: ten.1055/due south-2004-817915
PubMed Abstract | CrossRef Full Text | Google Scholar
Haasova, Chiliad., Warren, F. C., Ussher, Chiliad., Janse Van Rensburg, Chiliad., Faulkner, G., Cropley, Yard., et al. (2013). The acute furnishings of physical activity on cigarette cravings: systematic review and meta-assay with individual participant information. Addiction 108, 26–37. doi: ten.1111/j.1360-0443.2012.04034.ten
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hillman, C. H., Erickson, K. I., and Kramer, A. F. (2008). Exist smart, exercise your center: exercise effects on brain and noesis. Nat. Rev. Neurosci. nine, 58–65. doi: 10.1038/nrn2298
PubMed Abstract | CrossRef Full Text | Google Scholar
Hirase, H., and Shinohara, Y. (2014). Transformation of cortical and hippocampal neural circuit by ecology enrichment. Neuroscience 280, 282–298. doi: x.1016/j.neuroscience.2014.09.031
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hollamby, A., Davelaar, Eastward. J., and Cadar, D. (2017). Increased physical fitness is associated with higher executive operation in people with dementia. Front. Public Heal. 5:346. doi: 10.3389/fpubh.2017.00346
PubMed Abstract | CrossRef Total Text | Google Scholar
Hötting, K., and Röder, B. (2013). Beneficial furnishings of concrete exercise on neuroplasticity and cognition. Neurosci. Biobehav. Rev. 37, 2243–2257. doi: x.1016/j.neubiorev.2013.04.005
PubMed Abstract | CrossRef Full Text | Google Scholar
Hötting, K., Schickert, North., Kaiser, J., Röder, B., and Schmidt-Kassow, Thou. (2016). The effects of acute physical exercise on memory, peripheral bdnf, and cortisol in young adults. Neural Plast. 2016, one–12. doi: 10.1155/2016/6860573
PubMed Abstract | CrossRef Full Text | Google Scholar
Hu, T., Zhou, F. J., Chang, Y. F., Li, Y. Due south., Liu, G. C., Hong, Y., et al. (2015). miR21 is associated with the cognitive improvement post-obit voluntary running wheel practice in TBI mice. J. Mol. Neurosci. 57, 114–122. doi: 10.1007/s12031-015-0584-viii
PubMed Abstract | CrossRef Full Text | Google Scholar
Huang, P., Fang, R., Li, B. Y., and Chen, S.-D. (2016). Practice-related changes of networks in aging and balmy cerebral harm brain. Forepart. Crumbling Neurosci. 8:47. doi: 10.3389/fnagi.2016.00047
PubMed Abstract | CrossRef Total Text | Google Scholar
Ieraci, A., Mallei, A., Musazzi, L., and Popoli, M. (2015). Concrete exercise and acute restraint stress differentially modulate hippocampal brain-derived neurotrophic factor transcripts and epigenetic mechanisms in mice. Hippocampus 25, 1380–1392. doi: 10.1002/hipo.22458
PubMed Abstract | CrossRef Total Text | Google Scholar
Isaacs, M. R., Anderson, B. J., Alcantara, A. A., Blackness, J. E., and Greenough, W. T. (1992). Do and the brain: angiogenesis in the adult rat cerebellum after vigorous physical activity and motor skill learning. J. Cereb. Blood Flow Metab. 12, 110–119. doi: 10.1038/jcbfm.1992.14
PubMed Abstract | CrossRef Full Text | Google Scholar
Ji, E. South., Kim, Y. M., Shin, M. S., Kim, C. J., Lee, Yard. S., Kim, G., et al. (2015). Treadmill exercise enhances spatial learning ability through suppressing hippocampal apoptosis in Huntington's disease rats. J. Exerc. Rehabil. 11, 133–139. doi: x.12965/jer.150212
PubMed Abstract | CrossRef Total Text | Google Scholar
Kleim, J. A., Cooper, North. R., and VandenBerg, P. M. (2002). Exercise induces angiogenesis but does not modify movement representations within rat motor cortex. Brain Res. 934, ane–6. doi: 10.1016/S0006-8993(02)02239-4
CrossRef Total Text | Google Scholar
Knapen, J., Sommerijns, E., Vancampfort, D., Sienaert, P., Pieters, One thousand., Haake, P., et al. (2009). Land anxiety and subjective well-being responses to acute bouts of aerobic exercise in patients with depressive and anxiety disorders. Br. J. Sports Med. 43, 756–759. doi: x.1136/bjsm.2008.052654
PubMed Abstract | CrossRef Total Text | Google Scholar
Korb, A., Bonetti, Fifty. Five., Da Silva, S. A., Marcuzzo, S., Ilha, J., Bertagnolli, One thousand., et al. (2010). Effect of treadmill exercise on serotonin immunoreactivity in medullary raphe nuclei and spinal cord following sciatic nerve transection in rats. Neurochem. Res. 35, 380–389. doi: ten.1007/s11064-009-0066-x
PubMed Abstract | CrossRef Full Text | Google Scholar
Kou, X., Li, J., Liu, X., Chang, J., Zhao, Q., Jia, Southward., et al. (2017). Pond attenuates d-galactose-induced brain crumbling via suppressing miR-34a-mediated autophagy impairment and abnormal mitochondrial dynamics. J. Appl. Physiol. 122, 1462–1469. doi: 10.1152/japplphysiol.00018.2017
PubMed Abstract | CrossRef Total Text | Google Scholar
Kramer, A. F., Hahn, Southward., Cohen, N. J., Banich, K. T., McAuley, E., Harrison, C. R., et al. (1999). Ageing, fitness and neurocognitive function. Nature 400, 418–419. doi: x.1038/22682
PubMed Abstruse | CrossRef Full Text | Google Scholar
Kronenberg, G., Bick-Sander, A., Bunk, E., Wolf, C., Ehninger, D., and Kempermann, Grand. (2006). Physical exercise prevents historic period-related reject in precursor prison cell activeness in the mouse dentate gyrus. Neurobiol. Crumbling 27, 1505–1513. doi: x.1016/j.neurobiolaging.2005.09.016
PubMed Abstract | CrossRef Full Text | Google Scholar
Lafenetre, P., Leske, O., Wahle, P., and Heumann, R. (2011). The beneficial furnishings of physical activeness on impaired adult neurogenesis and cognitive performance. Front end. Neurosci. 5:51. doi: 10.3389/fnins.2011.00051
PubMed Abstruse | CrossRef Full Text | Google Scholar
Lambourne, K., and Tomporowski, P. (2010). The effect of exercise-induced arousal on cognitive chore performance: a meta-regression analysis. Encephalon Res. 1341, 12–24. doi: x.1016/j.brainres.2010.03.091
PubMed Abstract | CrossRef Total Text | Google Scholar
Landers, D. K., and Arent, S. M. (2001). "Concrete action and mental wellness," in Handbook of Sport Psychology, eds R. N. Singer, H. A. Hausenblas, and C. Janelle (New York, NY: Wiley), 740–765.
Langdon, K. D., and Corbett, D. (2012). Improved working memory following novel combinations of concrete and cerebral activity. Neurorehabil. Neural Repair 26, 523–532. doi: 10.1177/1545968311425919
PubMed Abstruse | CrossRef Full Text | Google Scholar
Lees, C., and Hopkins, J. (2013). Effect of aerobic do on cognition, academic achievement, and psychosocial function in children: a systematic review of randomized command trials. Prev. Chronic Dis. ten:130010. doi: 10.5888/pcd10.130010
PubMed Abstract | CrossRef Full Text | Google Scholar
Lin, T. Due west., Shih, Y. H., Chen, S. J., Lien, C. H., Chang, C. Y., Huang, T. Y., et al. (2015). Running exercise delays neurodegeneration in amygdala and hippocampus of Alzheimer'southward disease (APP/PS1) transgenic mice. Neurobiol. Learn. Mem. 118, 189–197. doi: 10.1016/j.nlm.2014.12.005
PubMed Abstract | CrossRef Total Text | Google Scholar
Loprinzi, P. D., Frith, E., Edwards, K. Thou., Sng, E., and Ashpole, Northward. (2018). The furnishings of exercise on memory function among immature to center-aged adults: systematic review and recommendations for future enquiry. Am. J. Heal. Promot. 32, 691–704. doi: 10.1177/0890117117737409
PubMed Abstract | CrossRef Total Text | Google Scholar
Ludyga, Southward., Gerber, M., Brand, S., Holsboer-Trachsler, E., and Pühse, U. (2016). Astute furnishings of moderate aerobic practise on specific aspects of executive function in different age and fitness groups: a meta-analysis. Psychophysiology 53, 1611–1626. doi: 10.1111/psyp.12736
PubMed Abstract | CrossRef Full Text | Google Scholar
Maejima, H., Kanemura, N., Kokubun, T., Murata, K., and Takayanagi, K. (2018). Exercise enhances cognitive function and neurotrophin expression in the hippocampus accompanied by changes in epigenetic programming in senescence-accelerated mice. Neurosci. Lett. 665, 67–73. doi: ten.1016/j.neulet.2017.11.023
PubMed Abstract | CrossRef Total Text | Google Scholar
Mammen, G., and Faulkner, G. (2013). Concrete activeness and the prevention of low: a systematic review of prospective studies. Am. J. Prev. Med. 45, 649–657. doi: x.1016/j.amepre.2013.08.001
PubMed Abstract | CrossRef Full Text | Google Scholar
Mandolesi, L., Gelfo, F., Serra, 50., Montuori, S., Polverino, A., Curcio, Grand., et al. (2017). Environmental factors promoting neural plasticity: insights from animal and human studies. Neural Plast. 2017, 1–10. doi: 10.1155/2017/7219461
PubMed Abstract | CrossRef Total Text | Google Scholar
Marsh, H. W., and Sonstroem, R. J. (1995). Importance ratings and specific components of concrete self-concept: relevance to predicting global components of self-concept and exercise. J. Sport Exerc. Psychol. 17, 84–104. doi: 10.1123/jsep.17.1.84
CrossRef Total Text | Google Scholar
Meyer, J. D., Ellingson, L. D., Koltyn, K. F., Stegner, A. J., Kim, J. S., and Cook, D. B. (2016a). Psychobiological responses to preferred and prescribed intensity exercise in major depressive disorder. Med. Sci. Sports Exerc. 48, 2207–2215. doi: ten.1249/MSS.0000000000001022
PubMed Abstract | CrossRef Full Text | Google Scholar
Meyer, J. D., Koltyn, K. F., Stegner, A. J., Kim, J.-S., and Melt, D. B. (2016b). Relationships between serum BDNF and the antidepressant issue of acute exercise in depressed women. Psychoneuroendocrinology 74, 286–294. doi: 10.1016/j.psyneuen.2016.09.022
PubMed Abstract | CrossRef Full Text | Google Scholar
Molteni, R., Ying, Z., and Gómez-Pinilla, F. (2002). Differential furnishings of astute and chronic exercise on plasticity-related genes in the rat hippocampus revealed by microarray. Eur. J. Neurosci. 16, 1107–1116. doi: 10.1046/j.1460-9568.2002.02158.x
PubMed Abstract | CrossRef Total Text | Google Scholar
Niemann, C., Godde, B., Staudinger, U. M., and Voelcker-Rehage, C. (2014). Exercise-induced changes in basal ganglia book and cognition in older adults. Neuroscience 281, 147–163. doi: 10.1016/j.neuroscience.2014.09.033
PubMed Abstruse | CrossRef Full Text | Google Scholar
Nithianantharajah, J., and Hannan, A. J. (2006). Enriched environments, experience-dependent plasticity and disorders of the nervous organization. Nat. Rev. Neurosci. 7, 697–709. doi: x.1038/nrn1970
PubMed Abstruse | CrossRef Full Text | Google Scholar
Pahng, A. R., McGinn, M. A., and Paulsen, R. I. (2017). The prefrontal cortex as a critical gate of negative affect and motivation in alcohol utilise disorder. Curr. Opin. Behav. Sci. 13, 139–143. doi: 10.1016/j.cobeha.2016.11.004
PubMed Abstract | CrossRef Full Text | Google Scholar
Pan-Vazquez, A., Rye, Due north., Ameri, One thousand., McSparron, B., Smallwood, G., Bickerdyke, J., et al. (2015). Impact of voluntary exercise and housing weather on hippocampal glucocorticoid receptor, miR-124 and anxiety. Mol. Encephalon 8:40. doi: 10.1186/s13041-015-0128-8
PubMed Abstract | CrossRef Full Text | Google Scholar
Pedrinolla, A., Schena, F., and Venturelli, 1000. (2017). Resilience to Alzheimer'southward disease: the role of physical activity. Curr. Alzheimer Res. 14, 546–553.
Google Scholar
Penedo, F. J., and Dahn, J. R. (2005). Practise and well-beingness: a review of mental and physical health benefits associated with physical activity. Curr. Opin. Psychiatry eighteen, 189–193. doi: 10.1097/00001504-200503000-00013
PubMed Abstruse | CrossRef Total Text | Google Scholar
Pereira, A. C., Huddleston, D. East., Brickman, A. M., Sosunov, A. A., Hen, R., McKhann, G. 1000., et al. (2007). An in vivo correlate of do-induced neurogenesis in the developed dentate gyrus. Proc. Natl. Acad. Sci. UsA. 104, 5638–5643. doi: 10.1073/pnas.0611721104
PubMed Abstruse | CrossRef Full Text | Google Scholar
Petrosini, L., De Bartolo, P., Foti, F., Gelfo, F., Cutuli, D., Leggio, M. M., et al. (2009). On whether the environmental enrichment may provide cerebral and brain reserves. Brain Res. Rev. 61, 221–239. doi: 10.1016/j.brainresrev.2009.07.002
PubMed Abstract | CrossRef Full Text | Google Scholar
Raedeke, T. D. (2007). The relationship between enjoyment and affective responses to practise. J. Appl. Sport Psychol. 19, 105–115. doi: 10.1080/10413200601113638
CrossRef Total Text | Google Scholar
Richter, H., Ambrée, O., Lewejohann, L., Herring, A., Keyvani, 1000., Paulus, Due west., et al. (2008). Wheel-running in a transgenic mouse model of Alzheimer'due south disease: protection or symptom? Behav. Brain Res. 190, 74–84. doi: 10.1016/j.bbr.2008.02.005
CrossRef Full Text | Google Scholar
Robinson, A. Yard., Eggleston, R. L., and Bucci, D. J. (2012). Physical exercise and catecholamine reuptake inhibitors touch orienting behavior and social interaction in a rat model of attention-deficit/hyperactivity disorder. Behav. Neurosci. 126, 762–771. doi: x.1037/a0030488
PubMed Abstract | CrossRef Full Text | Google Scholar
Rodgers, W. M., Markland, D., Selzler, A. M., Murray, T. C., and Wilson, P. M. (2014). Distinguishing perceived competence and self-efficacy: an instance from exercise. Res. Q. Exerc. Sport 85, 527–539. doi: x.1080/02701367.2014.961050
PubMed Abstruse | CrossRef Full Text | Google Scholar
Saab, B. J., and Mansuy, I. Thousand. (2014). Neuroepigenetics of memory formation and impairment: the function of microRNAs. Neuropharmacology 80, 61–69. doi: 10.1016/j.neuropharm.2014.01.026
PubMed Abstract | CrossRef Full Text | Google Scholar
Sachs, 1000. L. (1981). "Running addiction," in Psychology of Running, eds M. H. Sachs and M. L. Sachs (Champaign, IL: Human Kinetics), 116–127.
Schwellnus, One thousand., Soligard, T., Alonso, J.-Grand., Bahr, R., Clarsen, B., Dijkstra, H. P., et al. (2016). How much is too much? (Part 2) International Olympic Committee consensus statement on load in sport and run a risk of illness. Br. J. Sports Med. l, 1043–1052. doi: 10.1136/bjsports-2016-096572
PubMed Abstruse | CrossRef Total Text | Google Scholar
Scully, D., Kremer, J., Meade, M. M., Graham, R., and Dudgeon, Chiliad. (1998). Physical exercise and psychological well being: a critical review. Br. J. Sports Med. 32, 111–120. doi: ten.1136/bjsm.32.two.111
PubMed Abstruse | CrossRef Full Text | Google Scholar
Serra, L., Cercignani, M., Petrosini, Fifty., Basile, B., Perri, R., Fadda, L., et al. (2011). Neuroanatomical correlates of cerebral reserve in alzheimer disease. Rejuvenation Res. xiv, 143–151. doi: x.1089/rej.2010.1103
PubMed Abstract | CrossRef Full Text | Google Scholar
Brusk, A. K., Yeshurun, S., Powell, R., Perreau, 5. Thou., Fox, A., Kim, J. H., et al. (2017). Practise alters mouse sperm small-scale noncoding RNAs and induces a transgenerational modification of male person offspring conditioned fear and anxiety. Transl. Psychiatry seven:e1114. doi: ten.1038/tp.2017.82
PubMed Abstract | CrossRef Total Text | Google Scholar
Sibley, B. A., and Etnier, J. Fifty. (2003). The human relationship betwixt physical activeness and cognition in children: a meta-analysis. Pediatr. Exerc. Sci. 15, 243–256. doi: ten.1123/pes.fifteen.three.243
CrossRef Full Text | Google Scholar
Sim, Y. J. (2014). Treadmill practice alleviates impairment of spatial learning ability through enhancing cell proliferation in the streptozotocin-induced Alzheimer's disease rats. J. Exerc. Rehabil. 10, 81–88. doi: x.12965/jer.140102
PubMed Abstract | CrossRef Full Text | Google Scholar
Snigdha, S., de Rivera, C., Milgram, N. W., and Cotman, C. W. (2014). Exercise enhances memory consolidation in the aging brain. Front. Aging Neurosci. 6:3. doi: 10.3389/fnagi.2014.00003
PubMed Abstract | CrossRef Full Text | Google Scholar
Steiner, B., Kronenberg, One thousand., Jessberger, South., Brandt, G. D., Reuter, K., and Kempermann, G. (2004). Differential regulation of gliogenesis in the context of adult hippocampal neurogenesis in mice. Glia 46, 41–52. doi: 10.1002/glia.10337
PubMed Abstruse | CrossRef Total Text | Google Scholar
Stessman, J., Hammerman-Rozenberg, R., Cohen, A., Ein-Mor, East., and Jacobs, J. M. (2009). Physical action, function, and longevity amid the very one-time. Arch. Intern. Med. 169, 1476–1483. doi: 10.1001/archinternmed.2009.248
PubMed Abstract | CrossRef Full Text | Google Scholar
Beau, R. A., Harris, A. B., Wiener, E. C., Dutka, M. V., Morris, H. D., Theien, B. E., et al. (2003). Prolonged exercise induces angiogenesis and increases cerebral blood volume in master motor cortex of the rat. Neuroscience 117, 1037–1046. doi: 10.1016/S0306-4522(02)00664-4
PubMed Abstract | CrossRef Full Text | Google Scholar
Thomas, S. N., Schroeder, T., Secher, N. H., and Mitchell, J. H. (1989). Cerebral blood catamenia during submaximal and maximal dynamic exercise in humans. J. Appl. Physiol. 67, 744–748. doi: 10.1152/jappl.1989.67.2.744
PubMed Abstract | CrossRef Full Text | Google Scholar
Thomson, D., Turner, A., Lauder, South., Gigler, Grand. E., Berk, L., Singh, A. B., et al. (2015). A cursory review of practice, bipolar disorder, and mechanistic pathways. Front. Psychol. 6:147. doi: 10.3389/fpsyg.2015.00147
PubMed Abstract | CrossRef Full Text | Google Scholar
Tiryaki-Sonmez, G., Vatansever, Due south., Olcucu, B., and Schoenfeld, B. (2015). Obesity, food intake and exercise: relationship with ghrelin. Biomed. Hum. Kinet. 7, 116–124. doi: 10.1515/bhk-2015-0018
CrossRef Full Text | Google Scholar
van Praag, H., Christie, B. R., Sejnowski, T. J., and Gage, F. H. (1999a). Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc. Natl. Acad. Sci. The statesA. 96, 13427–13431.
PubMed Abstruse | Google Scholar
van Praag, H., Kempermann, G., and Gage, F. H. (1999b). Running increases prison cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat. Neurosci. 2, 266–270.
PubMed Abstruse | Google Scholar
van Praag, H., Shubert, T., Zhao, C., and Cuff, F. H. (2005). Practice enhances learning and hippocampal neurogenesis in anile mice. J. Neurosci. 25, 8680–8685. doi: 10.1523/JNEUROSCI.1731-05.2005
PubMed Abstract | CrossRef Full Text | Google Scholar
Vatansever-Ozen, S., Tiryaki-Sonmez, 1000., Bugdayci, Grand., and Ozen, Grand. (2011). The effects of exercise on food intake and hunger: relationship with acylated ghrelin and leptin. J. Sports Sci. Med. ten, 283–291.
PubMed Abstract | Google Scholar
Vaynman, South., Ying, Z., and Gomez-Pinilla, F. (2004). Hippocampal BDNF mediates the efficacy of practise on synaptic plasticity and cognition. Eur. J. Neurosci. 20, 2580–2590. doi: x.1111/j.1460-9568.2004.03720.10
PubMed Abstruse | CrossRef Full Text | Google Scholar
Voss, M. Westward., Chaddock, L., Kim, J. South., VanPatter, 1000., Pontifex, M. B., Raine, L. B., et al. (2011). Aerobic fitness is associated with greater efficiency of the network underlying cerebral control in preadolescent children. Neuroscience 199, 166–176. doi: 10.1016/j.neuroscience.2011.ten.009
PubMed Abstruse | CrossRef Total Text | Google Scholar
Waddington, C. H. (1939). An Introduction to Modernistic Genetics. London: George Allen and Unwin Ltd.
Google Scholar
Wang, J., Fan, Y., Dong, Y., Ma, Thou., Ma, Y., Dong, Y., et al. (2016). Alterations in encephalon structure and functional connectivity in alcohol dependent patients and possible association with impulsivity. PLoS One 11:e0161956. doi: 10.1371/journal.pone.0161956
PubMed Abstract | CrossRef Total Text | Google Scholar
Weinberg, R. S., and Gould, D. (2015). Foundations of sport and exercise psychology, 6th Edn. Champaign, IL: Homo Kinetics.
Google Scholar
Windle, G., Hughes, D., Linck, P., Russell, I., and Woods, B. (2010). Is exercise effective in promoting mental well-being in older age? A systematic review. Aging Ment. Wellness 14, 652–669. doi: 10.1080/13607861003713232
PubMed Abstract | CrossRef Full Text | Google Scholar
Winter, B., Breitenstein, C., Mooren, F. C., Voelker, Thou., Fobker, M., Lechtermann, A., et al. (2007). High impact running improves learning. Neurobiol. Learn. Mem. 87, 597–609. doi: 10.1016/j.nlm.2006.11.003
PubMed Abstruse | CrossRef Total Text | Google Scholar
World Health Organization (2010). Global Recommendations on Physical Activity for Health. Geneva: WHO Press.
Yaffe, Chiliad., Fiocco, A. J., Lindquist, Grand., Vittinghoff, East., Simonsick, E. M., Newman, A. B., et al. (2009). Predictors of maintaining cerebral office in older adults: the Wellness ABC study. Neurology 72, 2029–2035. doi: 10.1212/WNL.0b013e3181a92c36
PubMed Abstract | CrossRef Full Text | Google Scholar
Young, S. Due north. (2007). How to increase serotonin in the human brain without drugs. J. Psychiatry Neurosci. 32, 394–399.
PubMed Abstract | Google Scholar
Zamani Sani, S. H., Fathirezaie, Z., Make, Southward., Pühse, U., Holsboer-Trachsler, E., Gerber, M., et al. (2016). Physical activity and self-esteem: testing direct and indirect relationships associated with psychological and physical mechanisms. Neuropsychiatr. Dis. Treat. 12, 2617–2625. doi: 10.2147/NDT.S116811
PubMed Abstract | CrossRef Full Text | Google Scholar
Zanetti, M., Shigaeff, Northward., Menzes, A. H. T., and Takahashi, A. A. (2017). Cognitive reserve: bear witness of delayed of dementia - a case report. J. Dement. 1:101.
Zubala, A., MacGillivray, S., Frost, H., Kroll, T., Skelton, D. A., Gavine, A., et al. (2017). Promotion of physical activeness interventions for community domicile older adults: a systematic review of reviews. PLoS ONE 12:e0180902. doi: 10.1371/journal.pone.0180902
PubMed Abstract | CrossRef Total Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fpsyg.2018.00509/full
0 Response to "Peer Reviewed Articles on Why Exercise Is Important"
Post a Comment